Day 2 :
Keynote Forum
Marek Malecki
Phoenix Biomolecular Engineering Foundation, USA
Keynote: HIV universal vaccine
Time : 09:00-09:40

Biography:
Marek Malecki, M.D., Ph.D., is President of the Phoenix Biomolecular Engineering Foundation, San Francisco, CA, USA, and Principal Investigator on the Program Project, at the National Institutes of Health, Bethesda, MD, USA. He is a physician specialized in Oncology. He is a Visiting Professor at the University of Wisconsin, St James School of Medicine, and Medical Center for Postgraduate Training of Physicians, while teaching Oncology, Pharmacology, and Pharmacogenetics/Pharmacogenomics. He was elected by the faculty and students to the Rho Chi Honor Society for Excellence in Teaching and as the Faculty Role Model. Professor Malecki is the Editor-in-Chief of Journal of Stem Cell Research and Therapy, Journal of Genetic Syndromes and Gene Therapy, British Journal of Biotechnology, Section Editor at the Clinical and Translational Medicine, and Editor in Frontiers in Genetics, Frontiers in Bioengineering and Biotechnology, Journal of Cancer research and Therapy. He is a member of American Medical Association and American Society for Medical Genetics.
Abstract:
For many deadly viruses, there are no preventive and/or therapeutic vaccines approved by health authorities’ world-wide (e.g., Human Immunodeficiency Virus). But for some viruses prophylactic vaccines are very effective (e.g. Hepatitis B Virus). In this realm, we design, manufacture, test, and streamline in to the clinics novel viral universal vaccines (VUV). VUV have such unique features, that medical vaccination or natural infection induced immunity against some viruses (e.g., Hepatitis B Virus or Human Papilloma) upon the VUV’s administration to the infected with other, different viruses patients, is redirected against these other, newly infecting viruses (e.g., Influenza Virus). In this realm, we have bio-molecularly engineered human immunodeficiency virus universal vaccines (HIVUV). As per the Institutional Review Board approval and in compliance with the Declaration of Helsinki, all healthy donors and patients were presented with the Patients’ Bill of Rights and provided Patient Informed Consent. All the procedures were pursued by the licensed medical doctors (MDs). By flow cytometry, immunoblotting, and nuclear magnetic resonance, we have demonstrated high efficacy of HIVUV to engage HBV immunized patients’ immune system against HIV. Administration of HIVUV to blood or lymph of the HIV+ patients resulted in rapid reduction of the HIV viremia down to undetectable. It also resulted in protection of populations of CD4+ cells against HIV cytopathic decline.
Keynote Forum
Frederic J Deschamps
University Hospital of Reims, France
Keynote: Vaccinations in working populations
Time : 09:40-10:20

Biography:
Frederic J Deschamps is a Medical doctor (Lille- France University in 1990). He completed his PhD in Occupational Toxicology in 1993. He was nominated Professor of Medicine in 2002. In the last 20 years he has improved the Department of Occupational Diseases of the University Hospital of Reims (Champagne County). From 1995, he manages the Regional Institute of Occupational Health. He belongs to the French National University College of Occupational Researches and Practioners. He has focused his work an occupational infectious diseases and health effects of low doses toxics with long term exposure.
Abstract:
Vaccinations are the key of current prophylactic measures for occupational risks. It concerns mainly hepatitis B, but also other vaccinations: diphtheria, polio, tetanus, flu. Few of those are actually important to protect determined groups of workers who could be exposed to infections in relationship with their jobs. Studies concerning the assessment of prevalence for vaccination’s coverage were conducted among a miscellaneous population of workers. The knowledge and the behaviors of workers towards vaccinations were also overseen. Low adherences to vaccinations, including for the high occupational risk exposure groups, were found. Only few workers stated that vaccinations are effective and safe, therefore positively associated with willingness to be vaccinated. Despite recommendations, the widespread use of vaccines in the working population clearly postpones the vaccines intakes. It seems desirable to promote a list of mandatory vaccinations regarding to job practices. Education about vaccinations to workers will also improve their behaviors towards its coverage.
Keynote Forum
Roberto Grau
Universidad Nacional de Rosario, Argentina
Keynote: Metal nanoparticles as a novel tool to fight microbial biofilm development and antibiotic-resistance emergency
Time : 10:20-11:00

Biography:
Roberto Grau completed his PhD from National University of Rosario in Argentina and obtained his Post-doctoral studies from The Scripps Research Institute (TSRI), Department of Experimental Medicine at San Diego, California, USA. He is a Pew Latin American Fellow (San Francisco, USA), a Fulbright International Scholar (Washignton DC, USA) and the Director of the Molecular Microbiology and Environmental Science Laboratory of the National Council of Scientific Research of Argentina (CONICET). He obtained many national and international awards, and published more than 30 papers in reputed journals.
Abstract:
Biofilms are three-dimensional structures that contains billions of genetically identical bacteria imbibed in a self-produced extracellular matrix, which shelter them from antibiotics. More than 85% of chronic and/or recurrent human infections are linked to bacterial biofilms, and every day the microbicide arsenal against them becomes more limited. Accordingly, nano-material science engineering is emerging as a promising alternative for reducing bioburden in healthcare facilities. Here, we present a novel TiO2-coated copper and silver nanoparticles (CuNPs and AgNPs, respectively) with enhanced photocatalytic and antibacterial properties. The activity of CuNPs and AgNPs (MNPs) was measured against planktonic and sessile forms of the relevant cystic-fibrosis related Pseudomona aeruginosa (CFPA) and methicillin-resistant Staphylococcus aureus (MRSA). The planktonic and sessile growth (measured as the final cellular yield at 600 nm and crystal violet staining, respectively) of both pathogens was severely inhibited by submillimolar concentrations of MNPs (95% of growth inhibition, p<0.01). The anti-biofilm effect was exerted at a genetic level as revealed by the downregulated expression produced by the MNPs on transcriptional β-galactosidase and gfp-fluorescence reporter fusions to genes involved in an extracellular matrix synthesis. MNP-treatment of pre-formed CFPA and MRSA biofilms dramatically accelerated their dissembling and cellular death without dispersal cell formation. Interestingly, the emergency of MNP-resistant CFPA or MRSA after two-weeks treatment with sub-MIC concentrations of the microbicide was significantly lower (p<0.01) than the emerged resistance after bacterial treatment with clinical antibiotics. The importance of the MNP treatment adoption as a safe alternative for reducing pathogenic bioburden will be presented.
Keynote Forum
MarÃa Rosa Chaig
BioquÃmica y BiologÃa Molecular, Argentina
Keynote: Aminoglycoside antibiotics and hearing loss
Time : 11:20-12:00

Biography:
Abstract:
Mutations in mitochondrial DNA (mtDNA) have been found to be associated with sensorineural hearing loss. As part of genetic screening program for hearing loss, we studied 40 patients with sensorineural deafness, whose cause might have been after aminoglycoside (ATB-AG), treatment. The affected and control subject's DNA fragments spanning the 12S rRNA gene or tRNASer(UCN) gene, that are associated with both aminoglycoside-induced and non-Syndromic hearing loss, were amplified and studied by PCR-RFLP. Three families have the homoplasmic 7444G> A mutation in the tRNAser (UCN) gene, the analysis of the mitochondrial genome in three family members did not detect any other pathology mutation. The clinical history shows one syndromic phenotype for matrilineal family. In the first family the muscle biopsy findings in the proband (III-5) and her mother (II- 5), show in the electronic microscopy (EM) and in the light microscopy (LM) multiple mitochondrial abnormalities in the striated muscle. These findings have been correlated with the values from Citocromo Oxidase/Citrate Synthase ratio, which indicated poor activity of the Citocromo Oxidase. The matrilineal pedigree clinical feature, and the molecular, biochemical and morphological studies, might indicate that this is a novel syndromic presentation of the 7444G>A mutation in Córdoba - Argentina. In the fourth family, the report of the clinical, genetic, and molecular characterization in two of their members, revealed the variable phenotype of hearing impairment including audiometric configuration. Mutational analysis of the mtDNA in these pedigrees showed the presence of non syndromic homoplasmic 12S rRNA A827G mutation, which has been associated with hearing impairment. The A827G mutation is located at the A-site of mitochondrial 12S rRNA gene which is highly conserved in mammals. It is possible that the alteration of the tertiary or quaternary structure of this rRNA by the A827G mutation may lead to mitochondrial dysfunction, thereby playing a role in the pathogenesis of hearing loss and aminoglycoside hypersensitivity. Although the 827A> G mutation in the 12S rRNA, is associated with haplogroup B, its prevalence ≥ 2%, does not eliminate its participation and association to ototoxicity by ATB-AG. In addition, it is necessary to know more about the mechanism by which ATB-AG induces hearing loss, in the presence of the 7444 G>A mutation in the tRNAser (UNC).
Keynote Forum
MarÃa Rosa Chaig
BioquÃmica y BiologÃa Molecular, Argentina
Keynote: Aminoglycoside antibiotics and hearing loss

Biography:
Abstract:
Mutations in mitochondrial DNA (mtDNA) have been found to be associated with sensorineural hearing loss. As part of genetic screening program for hearing loss, we studied 40 patients with sensorineural deafness, whose cause might have been after aminoglycoside (ATB-AG), treatment. The affected and control subject's DNA fragments spanning the 12S rRNA gene or tRNASer(UCN) gene, that are associated with both aminoglycoside-induced and non-Syndromic hearing loss, were amplified and studied by PCR-RFLP. Three families have the homoplasmic 7444G> A mutation in the tRNAser (UCN) gene, the analysis of the mitochondrial genome in three family members did not detect any other pathology mutation. The clinical history shows one syndromic phenotype for matrilineal family. In the first family the muscle biopsy findings in the proband (III-5) and her mother (II- 5), show in the electronic microscopy (EM) and in the light microscopy (LM) multiple mitochondrial abnormalities in the striated muscle. These findings have been correlated with the values from Citocromo Oxidase/Citrate Synthase ratio, which indicated poor activity of the Citocromo Oxidase. The matrilineal pedigree clinical feature, and the molecular, biochemical and morphological studies, might indicate that this is a novel syndromic presentation of the 7444G>A mutation in Córdoba - Argentina. In the fourth family, the report of the clinical, genetic, and molecular characterization in two of their members, revealed the variable phenotype of hearing impairment including audiometric configuration. Mutational analysis of the mtDNA in these pedigrees showed the presence of non syndromic homoplasmic 12S rRNA A827G mutation, which has been associated with hearing impairment. The A827G mutation is located at the A-site of mitochondrial 12S rRNA gene which is highly conserved in mammals. It is possible that the alteration of the tertiary or quaternary structure of this rRNA by the A827G mutation may lead to mitochondrial dysfunction, thereby playing a role in the pathogenesis of hearing loss and aminoglycoside hypersensitivity. Although the 827A> G mutation in the 12S rRNA, is associated with haplogroup B, its prevalence ≥ 2%, does not eliminate its participation and association to ototoxicity by ATB-AG. In addition, it is necessary to know more about the mechanism by which ATB-AG induces hearing loss, in the presence of the 7444 G>A mutation in the tRNAser (UNC).
- HIV Vaccines | Vaccine Research & Development | Human Vaccines - Infectious & Non Infectious Diseases | Veterinary Vaccines | Vaccines against Viral & Bacterial Diseases
Location: Hall - Linate

Chair
Chit Laa Poh
Sunway University, Malaysia
Session Introduction
Godwin W Nchinda
CIRCB, Cameroon
Title: Targeting conserved broadly neutralizing epitopes within HIV-1 envelope gp41 MPER as vaccine immunogens for seronegative partners of HIV-1 discordant couples
Time : 12:00-12:25

Biography:
Godwin Nchinda has his expertise in optimizing vaccine candidates against infectious diseases for preclinical and clinical evaluation in sub Saharan Africa. He has developed DNA, viral vectored and protein vaccines targeting the encoded HIV-1 vaccines to dendritic cells in situ. Dendritic cells are vital in initiating and regulating immune responses. He and his colleagues are pioneering a new generation of vaccines which are harnessing dendritic cells in situ to improve vaccine efficacy against infectious diseases. This strategy has been described in several peer reviewed publications with him as co-author.
Abstract:
Background: The membrane proximal external region (MPER) of HIV-1 envelope glycoprotein-41 (gp41) is targeted by several broadly neutralizing antibodies whose conserved linear epitopes are promising targets for vaccine design. However, a formidable challenge has remained the difficulty to design and deliver MPER based immunogens for the efficient induction of such broadly neutralizing HIV-1 specific antibodies (bnAb). This is mainly because the linear bnAb MPER epitopes are poorly accessible to the immune system. The overall objective of this study therefore was the development and validation of an RNA coliphage Qβ display system for efficient presentation of conserved bnAb epitopes to the immune system
Method: To overcome the challenge of effective presentation of MPER to the immune system we have selectively engineered the surface of the RNA coliphage Qβ to display a 51 aa consensus MPER peptide upon the surface of the phage particle. The expression cassettes were used for the production of QβMPER recombinant hybrid phages after transformation of HB101 strain.
Results: Specific recognition of some reported bnAb epitopes within MPER were confirmed in ELISA using the three recombinant QβMPER phages together with an MPER restrictive peptide as antigens and the bnAb 4E10, Z13e1, 2F5 and 10E8 as antibodies. Next the prevalence of MPER-specific antibodies was determined in plasma from long standing antiretroviral naïve HIV-1 infected participants of the CIRCB AFRODEC cohort. The greater majority (84%) of participants’ plasma showed MPER peptide specific reactivity with anti-MPER specific IgG antibody titers ranging from 200 to 409600 comparative to background IgG antibody titer. In immunogenicity studies in Balb/c mice the recombinant phages induced significantly high Anti-MPER-specific IgG antibody responses (P<0.04) in at least 60% of mice following three inoculations of each recombinant phage.
Conclusion: Thus, these novel recombinant QβMPER phages can be used to monitor MPER-specific immune responses in HIV-1 exposed or infected people. In addition the recombinant QβMPER phages could be used as immunogens either alone as demonstrated here in mice or in combination with other strategies for the induction of MPER specific immunity against HIV-1.
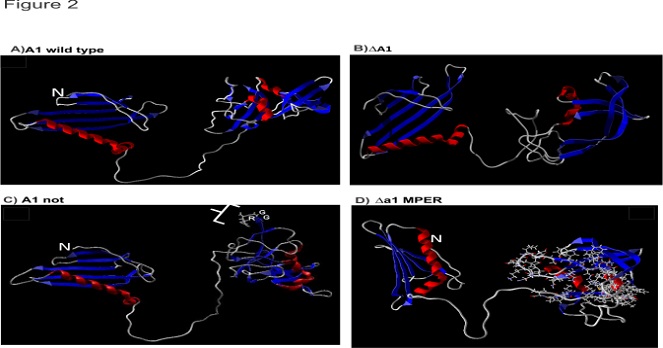
A) A1 wild type (B) ΔA1 (C) A1Not (D) ΔA1MPER (MPER highlighted in ball and stick rendering at the C terminus region of the protein). (N indicates the N terminus of the minor coat protein A1; red and blue colored structures indicate α-helix and β-sheets, respectively)
Xiao-Song He
VA Palo Alto Health Care System, USA
Title: Repeated influenza vaccination is a potential cause for reduced B cell response in the elderly
Time : 12:25-12:50

Biography:
Xiao-Song He has received his Doctorate degree from Fudan University, China and completed his Postdoctoral studies at Stanford University School of Medicine, USA. He is a Senior Research Scientist at VA Palo Alto Health Care System and Adjunct Professor at University of California in Davis, USA. He has published more than 60 original papers in the fields of Viral Immunology and Autoimmunity, and is a current member of the Editorial Advisory Board of The Journal of Infectious Diseases.
Abstract:
The disease burden of annual influenza epidemics is especially significant among elderly individuals. Although vaccination provides protection, influenza vaccine needs to be reformulated yearly due to the frequent mutations of circulating strains. Currently annual influenza vaccination is recommended for individuals aged six months or older. The protective efficacy of the inactivated influenza vaccine (IIV) is significantly lower in the elderly than the younger adults but the cause of this age effect is not fully understood. To address this issue, we investigated the circulating plasmablast response to IIV and found that the number of vaccine-induced plasmablasts, or activated B cells, was lower in the elderly than young adults. In addition, the numbers of de novo somatic hypermutations in the immunoglobulin genes of influenza-specific plasmablasts were also lower in the elderly, resulting in an antibody response poorly adapted to the new vaccine antigens. To further explore the cause of the reduced B cell response, we followed a cohort of individuals who received annual influenza vaccination in four consecutive years and measured their plasmablast response after each vaccination. The plasmablast response declined with increased number of vaccination, whereas the avidity of plasmablast-derived polyclonal antibodies did not increase with repeated immunization of the same influenza strain. Our findings suggest that repeated IIV immunization is a factor contributing to the reduced antibody response to influenza vaccination in the elderly who have extensive exposure to influenza antigens during their livetime. Therefore it is importance to develop universal influenza vaccines that do not require annual vaccination.
Mark Fife
The Pirbright Institute, UK
Title: Chicken IFITM gene knockout technology for enhanced vaccine production
Time : 13:40-14:05

Biography:
QTL and association studies), candidate gene mapping and molecular biology techniques. He has produced over 45 peer-reviewed publications and book chapters in this area before becoming a Group Leader at the Institute. His work has been the focus of extensive genome-wide and haplotype analysis using web-based SNP selector software that he has implemented at Pirbright. This work has culminated in the identification and characterization of several causal genes for important immune traits in chickens. His current research found a group of related genes called the interferon-inducible transmembrane (IFITM) genes that are able to prevent viruses from attacking and killing host cells. The aim of his group work is to determine the biology and genetic variation of these and similar immune genes in chickens; specifically, the ability of these genes to protect the host against avian viruses. The output of this work will be in identifying specific gene variants that correlate with resistance to a number of avian viruses, thus allowing poultry breeding programmes to select robust chickens, able to fight viral infections.
Abstract:
Type I interferon protect cells from viral infections through the induction of a group of genes collectively named interferon-stimulated genes (ISGs). Among these ISGs, are the IFITM (interferon-inducible transmembrane) which have been shown to restrict the replication of several highly pathogenic human viruses, including severe acute respiratory syndrome (SARS) coronavirus, filoviruses (Marburg virus and Ebola virus), influenza A viruses (IAVs), and flaviviruses (dengue virus). The genetics and genomics group have identified these antiviral proteins in the chicken (chIFITM) and have shown that a reduction in chIFITM expression results in an increase in the virus titre in CEFs infected with avian influenza A virus (AIV) H9N2, suggesting that chIFITMs have a functional role in the control of viral infections. The observation may have useful implications in terms of vaccine production. Many vaccines are produced in embryonated hen’s eggs or continuous avian cell lines. However, it is well established that the rate determining step in the manufacture of numerous vaccines is the induction of antiviral immune responses that prevents the replication of vaccine viruses. To generate chIFITM knock-down, we will use cutting edge genetic approaches such the CRISPR/Cas9 system which will directly target and knock-out chIFITM expression. We believe that this approach will overcome the rate limiting step in vaccine production, directly resulting in increased vaccine yields and improve the speed at which vaccines can be manufactured. We are currently in talks with major vaccine producers keen to adopt this internationally patented technology, to advance the field of both animal and human vaccine production. This work is being conducted in partnership with Horizon Discovery, using their extensive expertise in genetic modification using gene editing technologies. The broad objective of the project is to observe the effect the knock-out of chIFITM genes expression, achieved via CRIPSR/Cas9 transfection methods, has on viral titre in avian cell lines (commonly used for vaccine production) infected with influenza A virus. In addition, through analyzing the genetic material of a wide variety of chicken breeds and outlying avian species that differ in levels of resistance to these viruses, we hope to identify versions of these proteins that give protection, in laboratory, commercial and “backyard” chickens. Analysis of these proteins in the chicken presents opportunities not just for a greater understanding of viral resistance, but also as tools to combat viruses in the poultry farming. It may be feasible to selectively breed for birds with improved resilience to viral infections; however, this requires the identification of resistance-associated factors and knowledge of how they act.
Chit Laa Poh
Sunway University, Malaysia
Title: MicroRNA reduction of enterovirus 71 viral replication attenuates and confers protective immune response in mice
Time : 14:05-14:30

Biography:
Dr. Chit Laa Poh is a Distinguished Professor and Head of the Centre for Biomedical Sciences at Sunway University. She completed her PhD at Monash University (Australia) in 1980 and conducted short periods of postdoctoral training from the Pasteur Institute, Cambridge University and the University College London. She has previously worked in the Yong Loo Lin School of Medicine, National University of Singapore (NUS) for 25 years. She has published more than 85 papers in reputed journals and has been serving as an editorial board member of Journal of Biosceince and Bioengineering, Austin Journal of Tropical Medicine and Hygeine, Journal of Virology and Emerging Diseases and Annals of Translational Medicine and Epidemiology.
Abstract:
The hand, foot, and mouth disease (HFMD) is generally manifested as a subclinical infection, but fatal neurological complications can occur in young children. Epidemiological surveillance in China from 2008-2014 showed that 43.73% of HFMD cases were due to EV-A71. Up to date, there is no WHO-approved vaccine against EV-A71. This study demonstrated a novel miRNA vaccine construct for EV-A71 which decreased viral replication in vitro, whilst conferring a protective immune response in four-week old ICR mice. A vaccine construct was engineered to carry microRNA (miRNA) target sequences let-7a and miR-124a in the EV-A71 genome, allowing endogenous RNA silencing in specific cell types. The viral RNA copy number of the miRNA vaccine strain was much lower in RD (1.2 X 102) and SHSY-5Y cells (7.7 X 10) that expressed let-7a and miR-124a, respectively. The miRNA vaccine construct caused much reduction in plaque number (3.5 x 104 PFU/ml) in the SHSY-5Y cells as compared to the wild type virus (5.0 x 108 PFU/ml). No weight loss and hind limb paralysis were observed in the miRNA vaccine-administered mice (n=5) in comparison to the naïve group of mice (n=5). Significantly elevated systemic levels of IFN-γ and lower levels of pro-inflammatory cytokines TNF-α and IL-6 were detected. Higher CD8+ T cell response was elicited by the miRNA vaccine strain in mice as compared to the inactivated vaccine. The miRNA vaccine construct was able to confer protective immunity against EV-A71 sub-genotypes B3, C3 and C4. Ten serial passage studies to determine the genetic stability of the target sequences let-7a and miR-124a showed that the sequences did not revert to wild type virulence. Overall, the miRNA vaccine construct is an effective attenuation strategy for vaccine development.
